Payment for outcomes using GCP metrics is a way to significantly reduce clinical operations costs and increase value-for-money from your CRO or DCT vendor.
Payment for outcomes using GCP metrics shifts study costs from billable activities to measurable results. Professional service activities are hard to control. GCP metrics are hard to contest.
In this post, I suggest an alternative model for clinical data management, payment for outcomes using GCP metrics.
The notion of outcome-based-payments is not new and was discussed in the 2020 paper by Paula Lorgelly et al – Outcome-Based Payment Schemes: What Outcomes Do Patients with Cancer Value?
The context of the discussion was what outcomes patients value.
In this post, we will try and look the perspective of a sponsor – and identify what outcomes life science executives would value.
Current practice is paying for data entry pages in the EDC
Most sponsors who conduct clinical trials, use an EDC system. (Some sponsors still use a paper CRF, which is hard to grasp in this day and age).
Sponsors pay anywhere from $1,000 to $25,000 /monthly for an EDC system that enables users to transcribe from paper using online forms. Hybrid and decentralized clinical trials expand this model to data collection from patients using mobile ePRO and data collection from devices using an API.
Sites have SLO (service level objectives) to do data entry and are reimbursed for their data entry activities. This is a site cost, in addition to the cost of building and running an EDC system.
Curiously, the way DCT and hybrid trials are conducted today; neither patients nor devices have an SLO.
After paying for the EDC, you then pay for additional services for data managers, CRAs, SDV, on-site visits, and periodic reports. SDV and site monitoring visits, add another 30-50% to your total study cost. In additional to direct costs for clinical operations; there are indirect costs such as project management and users who leave in the middle of the project.
For example – what do you do if the data manager leaves the day before data lock?
Payment for outcomes – an alternative to paying for CRO and site activities
Data collection using EDC and patient ePRO should be a infrastructure service. This is user-generated content, where the users are site coordinators, patients and connected devices.
EDC and ePRO should be free.
The first step to a clinical data management, payment for outcomes model, is to make EDC and ePRO a free service. User-generated content is free for the users so EDC should be free for the users who collect data.
Project management should be free
If user-generated content from EDC, ePRO and devices is free, then it does not make sense to pay a monthly project management fee to a CRO for clinical data management.
A clinical data management, payment for outcomes model
The next step to a payment for outcomes model is to clearly identify the outcomes.
Although there will be difference clinical outcomes for each study, it seems to me that GCP provides an excellent framework for measuring and reimbursing clinical operations outcomes.
Clinical operations outcomes based in GCP
GCP has 3 pillars:
- Protocol compliance
- Patient safety
- Data quality
In DCT and hybrid trials, patient compliance to the protocol would be our first outcome for reimbursement.
Patient compliance outcomes
In decentralized and hybrid clinical trials, dosing and efficacy studies, patient compliance is crucial.
Question: How do you know how well the patients in the study are complying with the protocol?
Question: How do you enforce patient compliance during the critical washout period ?
In Figure 1 below, we see a case of a study using Flask Data EDC with an ePRO (patient reported outcome) module. We see a clear picture of ePRO compliance and treatment compliance. We can also see that the patients use the ePRO system consistently although one subject seems to have significant treatment compliance issues – missing 44 out of 72 capsules that they were supposed to take. Clearly ePRO data entry compliance does not insure treatment compliance.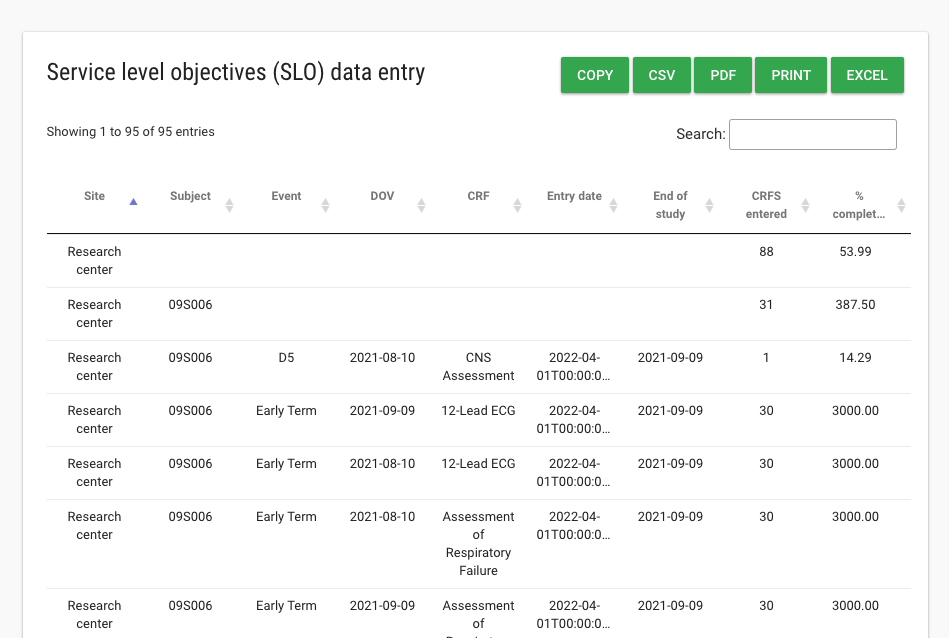
Figure 1
Data quality outcomes
In Figure 2 below – we take a sharp turn away from patient metrics and examine how well the study monitors are performing. As you can see in the case below, the site coordinators are extremely late on data entry. CRA site visits and SDV are occurring 3-4 months after patient visits. Some of the sites are entering future dates – which is a significant data quality issue.
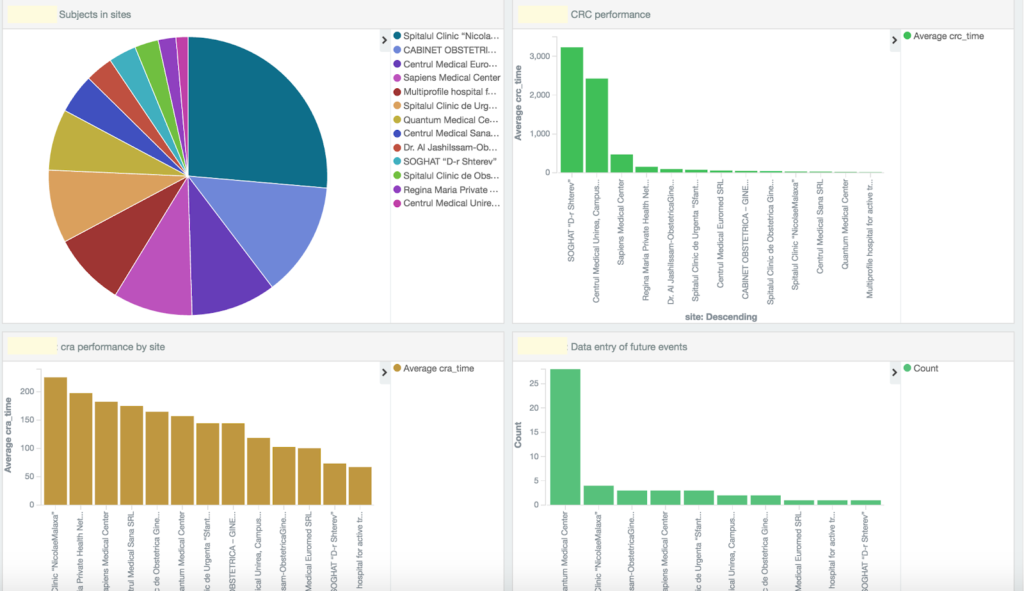
Figure 2
Patient safety outcomes – looking at DLT
In many dosing and efficacy studies (for example oncology), the protocol involves dosing titration over a period of time. The titration and march to DLT (dose-limited-toxicity) is closely related to patient safety.
How do we know how well we are doing with the dosing protocol? In Figure 3 below, we see that 55% of the patients were mis-dosed and were in violation of the study protocol placing the results of the entire study in jeopardy.
Figure 3
The future is payment for outcomes not for activities
Outcomes, not hours.
The EDC and ePRO systems are free.
Monitoring GCP metrics enables a payment for outcomes model
A process of automated monitoring that observes all your data (sites, patients, devices, users), all the time is enabling technology for monitoring GCP.
Payment for outcomes drives payment for value
For sure, big pharma has tools that provide analytics, automation and AI. No question at all.
If you have $500K for the EDC and another $300K for a RQBM solution, be our guest.
However – most sponsors (especially medical device, AI studies, and growth-stage biotech companies) require a cheaper and faster solution to fit their time-to-market goals and budget constraints.
Continuous improvement
The simplest version of a payment for outcomes model in clinical operations is to set a value for a target metric. For example – I will pay X for achieving patient compliance of 90-92% and 0.9X for achieving patient compliance of 80-89%.
However – we can amp it up and improve the contract to reward continuous improvement.
Since we are using metrics – it is easy to quantify improvement in a metric and compensate the vendor for improvement.
This requires Agility (learning as opposed to fixed process), Unlimited usage, and Speed.
Agility – Risk-based monitoring requires an initial risk-assessment. However, it is impossible to perform an effective risk-assessment/risk analysis without collecting data, and you need to run the study to collect data. In other words, monitoring needs to be cheap, agile and enable you to adapt to changes in data patterns and patient behavior.
An agile approach helps you discover issues across your entire data set, respond in real-time and refine your understanding of issues as the study progresses.
Unlimited usage – A SaaS subscription, driven by outcomes would need to be based on unlimited usage. Data management tools are self-service and included in the subscription.
The pricing should be accessible and start for less than a month of Starbucks coffee for your team.
Speed – Outcomes-based billing requires a pragmatic, agile and automated approach to monitoring.
You should expect a set of best-practice metrics that address key issues of GCP compliance metrics: quality, safety and protocol compliance:
For example one might expect a set of outcome metrics like this:
1. Dropouts/early termination rates
2. Adverse events/severe adverse event rates
3. CRC time – how long does it take for sites to enter data for a visit
4. CRA time – how long does it take for study monitors to perform document review
5. Concomitant medications and how they relate to AE, SAE
6. Query volumes and close rates
7. Patients with unsigned informed consent and in study
8. Failed inclusion/exclusion criteria and participating in study
Specific data models and study protocols enable you to introduce additional study-oriented metrics such as blood tests and hemoglobin levels in women. Studies with dosing titration can introduce an automated dosing compliance tool that enables you to see how the sites comply with the dosing protocol and whether they over or under-dose for their subjects. Decentralized clinical trials can track patient compliance in real-time.
Scale – An outcome model would enable scaling the clinical operations without requiring the sponsor to pay for additional infrastructure.
Payment for outcomes is payment for value
Payment for outcomes is an automated and agile approach to clinical data management.
The technology does the grunt work of observing the data and generating events, enabling you get an immediate picture of your data and your outcome metrcs.
Payment for outcomes is a high-value alternative to people and paper processes that enables you to focus on resolving issues in real-time, reduce avoidable rework and shorten the time to statistical report with cleaner data.
Our partners say it best:
“Flask Data is a novel and important tool for reinforcing patient adherence” –
Gabriel Veridze, former senior Parexel North America executive, CEO Prometheus World CRO
Contact us today for a free expert consultation on clinical trial success with Flask Data




